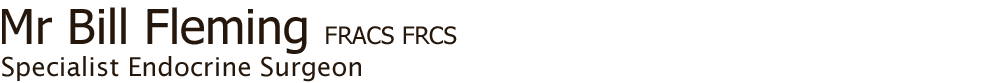Thyroid nodules
 Fig. 1: Thyroid nodule visible in the right lobeA thyroid nodule is a lump in an otherwise normal thyroid gland (Fig. 1). Thyroid nodules are very common, although most cannot be felt and are picked up on imaging. Epidemiological studies show that these palpable nodules are found in about 5% of women and 1% of men.
Fig. 1: Thyroid nodule visible in the right lobeA thyroid nodule is a lump in an otherwise normal thyroid gland (Fig. 1). Thyroid nodules are very common, although most cannot be felt and are picked up on imaging. Epidemiological studies show that these palpable nodules are found in about 5% of women and 1% of men.
However the overall incidence is much higher, with up to 65-70% having subclinical (undetected) thyroid nodules on ultrasound, with a greater incidence in women and the elderly.
Overall, incidental thyroid nodules are found in greater than 50% of neck ultrasounds, 16% of neck CT scans and 4% of PET scans.
Thyroid nodules tend to cover the full spectrum from completely solid lumps to completely fluid-filled lumps (cysts). Most patients have a variety of nodules in their thyroid, with combinations of mainly solid and mainly cystic lumps, but also nodules which have both features.
Nodules are divided into 5 pathological groups with distinctive histological features: hyperplastic nodules, colloid nodules, cysts, inflammatory nodules (seen in lymphocytic thyroiditis) and neoplastic nodules (either benign or malignant).
It is important to emphasise that less than 3% of these nodules are malignant (cancerous). If proven to be malignant however, these cancers are usually well differentiated and have a very good prognosis - more information about thyroid cancer can be found here.
Presentation
Most thyroid nodules are asymptomatic (cause no clinical problems). Thyroid nodules can present as a palpable lump or can be felt by the patient as a 'lump in the throat', particularly when swallowing. In rare cases a thyroid nodule can present with pain, usually due to sudden enlargement from bleeding which stretches the capsule of the thyroid. These bleeds have a characteristic history: the sudden onset of pain in the neck with the appearance of a lump or enlargement of a pre-existing lump, followed by easing of the pain and the lump getting smaller again over a few weeks as the blood is reabsorbed.
A nodule can also produce too much thyroid hormone (hot nodule or toxic nodule) and produce symptoms of thyrotoxicosis (see Thyrotoxicosis).
Patients presenting with thyroid nodules need a number of investigations, firstly to determine the thyroid function, and secondly to establish whether the lump is benign or malignant (cancerous).
Patients with incidental thyroid nodules found on a PET scan (PET-positive thyroid nodules) carry around a 60% risk of malignancy, of which >80% are papillary cancers, so all should undergo FNA cytological assessment (needle biopsy), and there should be a low threshold for surgical removal if there is any doubt.
All solitary nodules should be viewed with suspicion, particularly in children, who have a high incidence of malignancy (25%) in solitary nodules, with early lymphatic spread. Palpable dominant nodules in a multinodular goitre (that is single large lumps in an enlarged thyroid) have a slightly lower incidence of thyroid cancer than true solitary nodules. Palpable cystic (fluid-filled) lesions are often degenerating thyroid nodules, with a risk of malignancy that is again a little less than true solitary nodules.
The most common causes of benign (non-cancerous) thyroid nodules are:
- adenomas (benign overgrowths of normal thyroid tissue)
- cysts (fluid-filled lumps)
- Hashimoto's Thyroiditis
- less commonly, other forms of thyroiditis
Investigation
Patients who present with palpable or suspected thyroid nodules should undergo ultrasound investigation as the best first step in evaluating thyroid disease. For those found to have thyroid nodules incidentally on CT or PET scanning of the neck, ultrasound should also be performed to accurately determine the thyroid anatomy and the nature of the nodule(s).
In conjunction with the ultrasound, blood tests should be performed to determine thyroid function, and I would generally recommend that parathyroid function (calcium, parathyroid hormone and vitamin D estimations) should also be measured, as 15% of patients with thyroid disease will also have parathyroid disease.
There is no evidence that routine measurement of either serum calcitonin (looking for medullary cancer), or of serum thyroglobulin is of any benefit in the evaluation of thyroid nodules. Thyroglobulin is very non-specific and elevated in a number of thyroid diseases, both benign and malignant.
The next test should generally be fine needle aspiration cytology (FNA) with certain provisos (see below).
In summary, investigations should be as follows:
- high-resolution ultrasound
- thyroid function tests (TSH as a minimum, with T4 +/- T3)
- parathyroid function
- FNA cytology of suspicious nodules, >1cm nodules
- nuclear scan (only if thyroid function is abnormal)
1. Ultrasound
 Fig. 2: Ultrasound of a typical thyroid noduleDiagnostic ultrasound of the thyroid and cervical lymph nodes should be undertaken in all patients with a suspected thyroid nodule, nodular goitre or with an abnomality of the thyroid seen on another type of scan, such as a CT or PET scan.
Fig. 2: Ultrasound of a typical thyroid noduleDiagnostic ultrasound of the thyroid and cervical lymph nodes should be undertaken in all patients with a suspected thyroid nodule, nodular goitre or with an abnomality of the thyroid seen on another type of scan, such as a CT or PET scan.
A high-resolution ultrasound is performed to look at the nodule, although it can throw up more questions than answers at times. More than 20% of patients thought to have a single nodule on examination will have a second nodule on ultrasound, and 10% of patients with a palpable nodule will have nothing found on ultrasound.
An ultrasound scan cannot give a definitive answer as to whether a nodule is benign or malignant, but it can show characteristic markings of both, and show if the nodule is a cyst (Fig. 2). It can also identify the number of nodules, their size and position in the gland, the presence of lymphadenopathy, and if the nodules have any suspicious features suggesting potential malignancy.
The criteria that suggest malignancy of a nodule on ultrasound (Fig. 3) are:
- microcalcifications
- irregular borders
- marked hypoechogenicity
- extraglandular extension
- high blood flow within the nodule
- taller rather than wide shape
 Fig. 3: Ultrasound features of thyroid nodules and relative risk of malignancy (Source: 2015 American Thyroid Association Guidelines)
Fig. 3: Ultrasound features of thyroid nodules and relative risk of malignancy (Source: 2015 American Thyroid Association Guidelines)
Cystic (fluid-filled) thyroid swellings are less likely to be malignant than solid swellings, but cystic lesions in men are more likely to be malignant than in females. Krukowski's unit in Aberdeen published in 2008 that cysts that recur after aspiration, even with benign cytology, should undergo surgery, as 54% of patients with non-diagnostic or benign cytology were in fact cancerous.
2. Fine needle aspiration (FNA) cytology
The advent of fine needle aspiration cytology (FNA) more than 30 years ago significantly improved our ability to diagnose and appropriately treat thyroid nodules, and led to a more than 30% reduction in the number of operations for benign disease in the 1980s. Although ultrasound and nuclear scanning can give a probability that a lump is benign or malignant, only a biopsy can truly answer the question in any definitive way.
In this test, a very fine needle (25-27 gauge) is inserted into the nodule and a few cells removed for the cytologist to study under a microscope. The cytologist then determines whether the nodule is benign or malignant (Fig. 4). Fig. 4: Ultrasound guided FNA thyroid cytology
Fig. 4: Ultrasound guided FNA thyroid cytology
Cytology is relatively painless, can be performed with or without local anaesthesia, and ideally should be performed under ultrasound guidance. This ensures precise needle placement and therefore reduces unsatisfactory (or non-diagnostic) samples and improves diagnostic accuracy.
Ultrasound guidance is very important in the case of fluid-filled (cystic) lumps. Invariably the fluid contents will give no hint of malignancy, but biopsy of a thickened cyst wall under ultrasound control may identify an otherwise unrecognised thyroid cancer.
FNA should generally be confined to nodules >1cm in size, as they have the greater potential to be clinically significant cancers. Occasionally nodules <1cm should be biopsied if they have a suspicious ultrasound appearance, or if there is associated lymphadenopathy (enlarged lymph glands in the neck).
Diagnostic accuracy of an FNA of a suspicious lymph node is enhanced by not only looking at the cytology, but also by sending an aspirate for thyroglobulin estimation. Lymph nodes should not contain thyroglobulin, so if it is present this suggests that the node contains a metastasis from a thyroid cancer.
FNA is not recommended for overactive or 'hot' nodules, seen on a thyroid nuclear scan. These are rarely malignant and they will often show cell changes that mimic thyroid cancer, resulting in a misleading diagnosis and perhaps an unnecessary operation.
The results of thyroid FNA should always be interpreted in the light of the clinical findings and if the cytology does not tally with what has been found on examination then the FNA should be repeated, or consideration given to removing the lump. Clinically suspicious nodules should really be regarded as malignant despite negative cytology, until histological diagnosis has been confirmed.
Cytology allows accurate diagnoses to be made in colloid nodules and inflammatory thyroid disease (Hashimoto’s, De Quervain’s and tuberculosis). Papillary, medullary, anaplastic cancers and thyroid lymphomas are diagnosed with ease, but the diagnosis of follicular cancers is virtually impossible as cellular changes alone are insufficient to make the diagnosis. If the pathology is bizarre, medullary cancer must be excluded by staining for calcitonin and serum calcitonin analysis. It is best to treat all suspicious cytology surgically.
The classification of the results of fine needle aspiration is complex and has usually been interpreted as either non-diagnostic, benign, suspicious (atypical) or malignant. The latest guidelines from the American Thyroid Association recommend the more structured Bethesda reporting system for thyroid cytology, developed by the National Cancer Institute in Bethesda, USA.
The results of the cytology in the Bethesda classification can be:
Non-diagnostic or unsatisfactory (risk of malignancy: 1-4%): Inadequate numbers of cells have been aspirated, so the FNA should be repeated. If still non-diagnostic, then the management depends on clinical suspicion, patient wishes & ultrasound appearance (see sections below on 'Should FNA be repeated?' and 'FNA Sampling Errors').
Benign (or not malignant) (risk of malignancy: 0-3%): These are colloid or hyperplastic (adenomatous) nodules, or thyroiditis, and are very low risk lesions. These can usually be followed up clinically, with or without repeated FNA, providing there are no other risk factors (Fig. 5). In high risk cases, thyroid lobectomy is justified.
Is size of nodule important? It is still unclear from the evidence whether patients with nodules >4cm in size, and benign cytology, carry a higher risk of malignancy and should be managed differently than those with smaller nodules.
 Fig. 5: Benign cytology of colloid goitre
Fig. 5: Benign cytology of colloid goitre
Indeterminate / Atypia or follicular lesion of undetermined significance (AUS/FLUS) (risk of malignancy: 5-15%): Some 3 to 6% of thyroid FNAs are not easily classified into the benign, suspicious, or malignant categories. The AUS/FLUS group caters for this small minority of FNA results, and should be used sparingly. Such a result should trigger a close follow up regimen, with repeated FNA at 3 months. If there are other high risk factors, it is safer to remove the lesion by hemithyroidectomy.
Follicular neoplasm (inc. Hurthle cell neoplasm - HCN) (risk of malignancy: 15-30%): This result is usually because of a follicular lesion or suspected follicular cancer, which cannot be determined by FNA (Fig. 6). Typically these lesions have high cellularity, may contain an abundance of Hurthle cells, and colloid is scant or absent. The hallmark however, is a disturbed cytoarchitecture: follicular cells are arranged predominantly in microfollicular or trabecular groups, cellular crowding and overlapping are conspicuous, and the follicular cells are usually larger than normal. These lesions should be removed (usually by a hemithyroidectomy) and subjected to formal histology.
Higher risk group for malignancy, with a follicular lesion result, is a male patient, with a solitary nodule >4cm in size.
 Fig. 6: Atypical cytology of a follicular neoplasm
Fig. 6: Atypical cytology of a follicular neoplasm
Suspicious for malignancy (risk of malignancy: 60-75%): If only 1 or 2 characteristic features of papillary cancer are present, if they are only focal and not widespread throughout the follicular cell population, or if the sample is sparsely cellular, a malignant diagnosis cannot be made with certainty. Such cases occur with some regularity, and they are best classified as “suspicious for malignancy,” or qualified as “suspicious for papillary carcinoma.”
Nodules called suspicious for papillary carcinoma are usually treated the same way as a confirmed cancer, by total thyroidectomy (occasionally by hemithyroidectomy), and usually with central nodal dissection. Most (60%-75%) prove to be papillary carcinomas, and the rest are usually benign follicular adenomas.
Malignant (risk of malignancy: 97-99%): This occurs in 3-7% of FNAs when the cytology is diagnostic of thyroid cancer (Fig. 7). The patient will generally be recommended to have surgery, usually total thyroidectomy and central neck dissection, but there are exceptions in patients with very low risk microcancers, and in those who have medical conditions that preclude surgery or those who have a limited life-span. The extent of surgery depends on the type of tumour, its size and the tumour staging.
 Fig. 7: Cytology of a papillary cancer of thyroid
Fig. 7: Cytology of a papillary cancer of thyroid
Should FNA be repeated?
The 2015 ATA thyroid cancer guidelines recommend that if the FNA is non-diagnostic it should be repeated, probably after three months, although this waiting time can be shortened if the ultrasound is particularly suspicious. The malignancy rates for nodules with two consecutive non-diagnostic FNA results has been reported as high as 14%. A repeat FNA, for patients with a single initial non-diagnostic result, yields a diagnostic result in about 60-70% of patients, but a third try after two non-diagnostic results is less likely to be successful.
Combining the non-diagnostic FNA result, with the ultrasound features of the nodule that might suggest malignancy, is the best approach to decide whether a nodule needs surgery. If there is repeated non-diagnostic results but US appearance is benign, then close observation or diagnostic hemithyroidectomy is appropriate.
However, if in addtion to the non-diagnostic FNA, there are suspicious ultrasound features, clinical features of malignancy, or the nodule has grown more than 20% during surveillance, then surgery should be considered.
FNA Sampling errors?
If there is initially benign cytology and it tallies with the clinical findings, there is a very low risk of misdiagnosis. However, a recent study found that repeating the FNA may change a benign result to a malignant/suspicious one in more than 13% of patients. In the majority of cases, the repetition corrected wrong cytological interpretation of results other than colloidal goitre, especially Hashimoto's thyroiditis and regressive changes. They suggested that repeating FNA in patients with benign cytology at about one year can reduce the rate of undiagnosed tumours.
It is also important to remember that there is a more than 10% false-negative rate in benign FNA results in nodules >4cm in diameter (that is the result is called benign but the nodule is actually malignant). This is due to the heterogeneity of some nodules, which can harbour malignant cells in an area of the nodule which has NOT been sampled.
Similarly choosing which of several nodules to biopsy can alter the outcome. In general, it is better to sample the largest nodule, and a recent study has shown if you biopsy the larger of 2 nodules there is an 86% detection rate. If however, you biopsy the larger of 3 nodules, this drops to a 50% detection rate.
The choice of which nodule to biopsy should not rely on size criteria alone, but should also take into account other US features of malignancy, such as invasion or microcalcification for example. The malignant nodule may not necessarily be the largest one.
3. Nuclear medicine scan
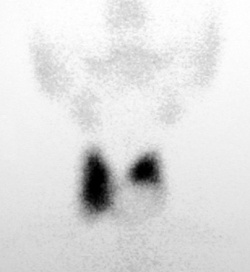 Fig. 8: Thyroid nuclear scan with 'cold' noduleIf there is evidence of thyrotoxicosis (overactivity) on the thyroid function tests then a nuclear medicine scan, either radioactive iodine or technetium scan, is indicated to determine the cause. This will discriminate between Graves’ disease, toxic multinodular goitre and a toxic thyroid nodule (Plummer’s disease).
Fig. 8: Thyroid nuclear scan with 'cold' noduleIf there is evidence of thyrotoxicosis (overactivity) on the thyroid function tests then a nuclear medicine scan, either radioactive iodine or technetium scan, is indicated to determine the cause. This will discriminate between Graves’ disease, toxic multinodular goitre and a toxic thyroid nodule (Plummer’s disease).
There is little benefit to be gained in performing thyroid nuclear scan in patients who have normal thyroid function, as it will not sort out benign and malignant nodules.
Nodules detected by radioactive iodine scans are classified as cold (80%), warm (15%) or hot (5%). If a nodule is not producing thyroid hormones, or is predominantly cystic, then it will not take up significant amounts of radioactive iodine and will appear 'cold' on the scan.
Cold nodules however, are only 1-2% more likely to be malignant, so nuclear scan is not helpful in determining the need for surgery.
A nodule that is actively producing hormone will take up the radioactive iodine and appear darker or 'hot' on the scan. Almost all 'hot' nodules are benign and do not require FNA cytology, as it can be misleading because of the cellular changes of overactivity (Fig. 8).
Surgery for thyroid nodules
So, what should be done with these thyroid nodules? In the case of malignant nodules the answer is generally easy, with most recommended for surgery. This will usually involve total thyroidectomy and central node dissection on the side of the tumour, but occasionally hemithyroidectomy may be appropriate in very low risk cancers. Further information about thyroid cancer can be found here.
In the case of benign (non-cancerous) nodules, the answer becomes more complex. True solitary nodules are always viewed with more suspicion, but provided the FNA and imaging are unequivocally benign these nodules can be watched and reviewed on a regular basis. Many patients however, request removal simply for peace of mind, and usually this requires hemithyroidectomy (removal of the whole of one side of the thyroid). True solitary nodules in children and younger men should also only be watched with a high index of suspicion.
The minimum surgery performed is a hemithyroidectomy, and many patients ask why not just take the nodule out? The problem is that operations to just remove the nodule carry a higher complication rate, a high risk of recurrence and the downside of being inadequate treatment if it turns out to be an unexpected cancerous nodule. Going back at redo surgery to remove the remainder of the thyroid on a side with previous partial thyroidectomy (nodule removal only) carries a 10x greater risk to the voice from having to operate in a scarred area.
On the other hand there is no greater risk to the recurrent laryngeal nerve if the side being operated on has been untouched previously, such as in the case of a completion thyroidectomy, necessary because of an unexpected cancer in the first side. That is why at a minimum, a complete hemithyroidectomy should be performed if nodule surgery is contemplated.
In the case of a dominant nodule in a sea of other nodules, the best treatment is determined by how extensive the disease has become and whether one side of the thyroid can be preserved. In this situation total thyroidectomy may be best to prevent an almost certain second operation in later life. However, if one side has only a few sub-centimetre sized nodules, then this side can usually be preserved to maximise the chances of avoiding the need for life-long thyroxine tablets.
Further information about the management of goitre can be found on the webpage Goitre.